Výzkum
Výzkum
Vedoucí oddělení:
RNDr. Dávid Drutovič, Ph.D.
|
|
|
Videospot laboratoře
Odkaz pro stažení videa a titulků.
Novinky
- Přednáška prof. Schindler
- Objev funkce tří Aurora kináz v savčích oocytech a reprodukci
- Chromozomy ve vajíčku řídí své vlastní rozdělení
Činnost laboratoře
Život všech organizmů, včetně člověka, vyžaduje/klade důraz na integritu informace uložené v DNA. DNA v jádře buňky je organizovaná do chromozomů. Všechny chromozomy (spolu s mitochondriální DNA) tvoří genom. Buňky disponují velmi sofistikovanými mechanizmy, které rozpoznávají poškození DNA a indukují její opravu – říká se jim Odpověď na poškození DNA (z angl. DNA damage response (DDR). Oslabená integrita DNA je úzce spojená s rakovinou. Avšak, oslabená integrita DNA má také přímé spojení k onemocněním, která nejsou rakovinová, jako jsou například pohlavní neplodnost nebo neurologické choroby.
V naší Laboratoři se použitím technik kvantitativního fluorescenčního zobrazení na fixovaných i živých buňkách, snažíme pochopit, jak souvisí integrita genomu savčích oocytů s pohlavní neplodností a neurologickými onemocněními.
Naše Laboratoř je součástí Centra PIGMOD podporovaného z projektu ExAM.
Introduction to the group and its research
The live of all organisms including human insist on the integrity of information stored in DNA. DNA in the nucleus of the cell is organised into the individual chromosomes. All chromosomes (and mitochondrial DNA) form the genome. The cells possess very sophisticated mechanisms that recognize DNA damage and induce DNA repair – DNA damage response (DDR). Compromised DNA integrity is widely connected to cancer. However, compromised DNA integrity has also clear link to non-cancerous diseases such as infertility or neurological disorders.
In our group we use mainly quantitative fluorescence imaging techniques both in fixed and live cells to address the topic of genome integrity in mammalian oocytes in connection to infertility and neurological Huntington’s disease.
Our laboratory is an integral part of Pigmod Center supported by the project Experimental Animal Models (EXAM).
Research
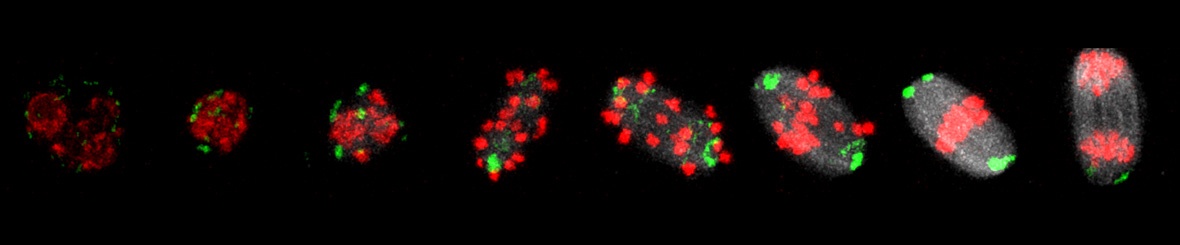
Chromosome dynamics and integrity in female meiosis
This research interest originates from the existence of the two greatly distinct cell lines in animals: germ cells and somatic cells. They have significantly different demands for the genetic stability. The germ lines must ensure transmission of the genetic information to the offsprings and must be very effective in the DNA integrity maintenance. In somatic cells, DNA repair is a compromise between growth speed, cancer risk and also the cost for DNA repair.
Until now a great effort was put into the research of chromosome segregation in female meiosis (oocytes). However, the correct chromosome segregation is the only one part of protection of the genome integrity. Also DNA repair of lesions such as double and single strand DNA breaks, crosslink DNA and cell cycle checkpoint outside anaphase are clearly very important, but largely unknown in germ line (oocytes and early embryos). DNA damage can arise from exogenous attacks such radiation but also from own basic metabolism (oxygen radicals) or DNA metabolism (replication, transcription).
In mammalian oocytes we focus both on the mechanism of chromosome segregation as well as on DNA damage response (DDR) on the DNA double strand breaks (DSB). Correct chromosome segregation is connected to normal cell cycle progression, normal spindle formation and functional spindle assembly checkpoint (SAC). We investigate signalling pathways that control these critical cellular processes. In the case of DSB we are trying to address the questions how are DSB recognized during meiotic maturation and how do oocytes deal with them? We are also interested in a potential link between DSB and whole chromosome segregation problem.
DNA damage response in neurological Huntington’s disease
The connection of DNA damage to cancer is well known. However neurological disorders, for example Huntington’s disease, are also associated with DNA damage and compromise DNA repair, although they have completely different impact for patients in comparison to cancer. Very interestingly, it is known from records that patients with Huntington’s disease have significantly lower cancer incidences. It provokes a question, how it is possible that DNA damage can lead to two completely different problems: cancer or neurodegeneration? We are interested in DSB response in cells compromised by the presence of mutated Huntingtin that is causative for Huntington’s disease. In this research topic we are working in close collaboration with the Laboratory of Cell Regeneration and Plasticity at our institute. This laboratory created transgenic minipig of Huntington’s disease. This model provides change to better address progressive character of Huntington’s disease (including DSB accumulation) in comparison to accelerated and short-time living mouse models such R6/2. Our goal is understand problems with DSB response in Huntington’s disease and help to use this knowledge both for diagnostic and therapeutic purpose.